Still the largest sector of the industry, monoclonal antibodies (MAbs) have dominated the biopharmaceutical stage for over 30 years. Some observers might think there’s nothing new to say about these molecules; others point to antibody derivatives as a more exciting alternative. But MAbs are far from an outdated technology. From biosimilar developments to cell-free synthesis to yeast display, immunogenicity improvements, and fully human antibodies — as well as improvements in process efficiency and cost reductions, as discussed herein — the…
Friday, October 30, 2020 Daily Archives
The Therapeutic Monoclonal Antibody Product Market
Commercial development of therapeutic monoclonal antibodies (MAbs) began in the early 1980s, and by 1986 the first MAb product had been approved in the United States: muromonab-CD3 (trade name Orthoclone OKT3, marketed by Janssen-Cilag) for prevention of kidney-transplant rejection. Since its approval, therapeutic MAbs and antibody-related products such as Fc-fusion proteins, antibody fragments, and antibody–drug conjugates (collectively referred to herein as “MAb products”) have grown to become the dominant product class within the biopharmaceutical market. They have been approved for…
Tracking Therapeutic Antibody Development in a Pandemic
The COVID-19 pandemic has generated a significant and rapid response from scientists who aim to develop drugs and vaccines in the academic, government, and industrial sectors. Such interventions are essential to containing SARS-CoV-2, the coronavirus that causes the COVID-19 disease. To inform and educate the public about global efforts to develop targeted therapies such as monoclonal antibodies (MAbs), The Antibody Society (TAS) and the Chinese Antibody Society (CAS) have designed and implemented a freely available online database called the COVID-19…
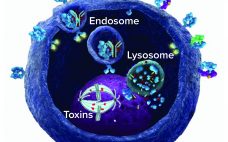
G-Protein–Coupled Receptors: Promising Targets for Antibody–Drug Conjugates
G-protein–coupled receptors (GPCRs) are a large and diverse family of seven-transmembrane–domain proteins expressed on the surface of human cells. These molecules respond to external stimuli by initiating signal-transduction pathways that affect the expression of a large family of genes — which, in turn, regulate a range of vital physiological processes and functions. Figure 1 illustrates the general pathways of GPCRs. Without these proteins, humans simply could not survive: Without β-adrenergic receptors, we could not regulate our blood sugar, for example;…
Sartorius to open Massachusetts customer interaction center
The facility will serve as a focal point for Sartorius’ US customers to test and discover bioprocess solutions for their biopharma projects. To be known as the Customer Interaction Center (CIC), the facility in Marlborough, Massachusetts will serve as a center of excellence for Sartorius. “The new site will be a focal point for customers in what is now one of the world’s largest development hubs for biopharmaceutical products,” the firm said. The 40,000 square-foot facility aims to optimize factory…
Lonza to support AstraZeneca’s COVID-19 antibody combo
Lonza will provide drug substance manufacturing for a potential COVID-19 monoclonal antibody combination therapy from its mid-scale facility in Portsmouth, New Hampshire. The deal, financials of which have not been disclosed, will see Swiss contract development and manufacturing organization (CDMO) Lonza make two monoclonal antibodies (mAbs) for AstraZeneca’s candidate AZD7442, currently in Phase I clinical studies as a potential treatment of COVID-19. The project will be one of the first from Lonza’s latest facility in Portsmouth, commissioned in 2018, with…
Novartis continues Kymriah capacity expansion with Japanese CDMO
Novartis has tripled production capacity for its commercial CAR-T therapy Kymriah over the past year and continues to ramp up through a deal with Japan’s Foundation for Biomedical Research and Innovation (FBRI). Kobe, Japan-based FBRI has received marketing authorization from Japan’s Ministry of Health, Labor and Welfare (MHLW) to manufacture Kymriah (tisagenlecleucel) for Swiss pharma giant Novartis. The approval expands the manufacturing footprint of the chimeric antigen receptor (CAR) T-cell therapy, bringing Novartis its first Asian production facility. Furthermore, according…