Hanns-Christian Mahler (head of drug product services, Lonza), BPI Theater @ BIO, June 8, 2016 11:20–11:40 am A drug product transfers a biomolecule (drug substance) into a form that will be usable by patients. It contains the active ingredient with excipients and can include packaging or a device for administration. In many cases, patients must be able to administer drugs to themselves. “Think about how the drug will be administered before designing your formulation,” Mahler suggested. Keeping a biomolecule intact…
Thursday, August 11, 2016 Daily Archives
The Portrait of a Protein: Exposing Its Character with Advanced Analytics
Greg Adams (director of analytical and formulation development, FUJIFILM Diosynth Biotechnologies), BPI Theater @ BIO, June 8, 2016 11:40 am –12:00 pm Fujifilm Diosynth Biotechnologies is a global contract development and manufacturing organization (CDMO) that has worked on multiple molecules. Adams said that proteins are complicated molecules with three dimensional (3-D) structures (primary, secondary, and tertiary) that are apt to self-aggregate. Monoclonal antibodies (MAbs), which we think of as relatively simple proteins, actually have very complex 3-D structures, with multiple…
Benefits to Strategic Outsourcing of Biologics Analytical Studies
Michael Merges (director of Catalent Biologics), BPI Theater @ BIO, June 8, 2016, 1:40–2:00 pm Many small companies do not have the expertise or specialized technologies to do the necessary testing that underlies biologics manufacturing. Expertise is needed in microbiology, raw materials testing, characterization, and method development. At the same time, testing is taking place earlier in product development. Bioassays are the most outsourced service, and that is expected to continue. Catalent has over 25 years of experience in characterizing…
Insourcing, Outsourcing, and Everything in Between: Tough Questions, Real Answers, and How to Make the Correct Decision for Your Situation
Greg Beattie (corporate vice president for global insourcing solutions and research support services, Charles River Laboratories), BPI Theater @ BIO June 8, 2016, 2:00–2:20 pm When you think of Charles River Laboratories,” Beattie began by asking, “do you associate it with anything other than rodents?” The company is very well known for selling rats and mice. However, it is also one of the world’s largest outsourcers in the biological testing field. The company is also one of the largest “insourcing”…
Making the Correct Outsourcing Decisions
A BPI Theater Roundtable at the 2016 BIO Convention On Wednesday, 8 June 2016, Gil Roth (president of the Pharma and Biopharma Outsourcing Association, PBOA) chaired a lunchtime roundtable titled, “Making the Correct Outsourcing Decisions.” He brought together a panel of four experts: Cory Lewis (vice president of business development and marketing at Cook Pharmica) Andrew Sanford (vice president of global business development for biologics at Catalent) David Powell (Pfizer CentreOne) Rajan Puri (senior director of business development at Therapure…
On-Demand Product Development Teams
A BPI Theater Roundtable at the 2016 BIO Convention On Wednesday, 8 June 2016, Joshua Speidel (managing director of the commercial practice team at the Latham Biopharm Group) chaired an afternoon roundtable titled, “On- Demand Product Development Teams.” He brought together a panel of four experts: Matt Rebold (director of business development at ICON Imaging and Technology — West Coast) Paul Jojorian (global head of technology transfer for Patheon Biologics) Eiry Wyn Roberts (vice president of research and development for…
CHOvolution™ Delivering Your Commercial Success: An Innovative Integrated Cell Line Development Platform
Celonic’s CHOvolution™ kit comes with a wide range of exceptional services and solutions, on top of high-performance cells. In addition to robust and highly efficient technology, we offer excellent technical support, including comprehensive protocols, audits, tailored workshops, and a 24-7 information access Internet-based platform with a hotline. For any drug developer or service provider, cell line development is one of the most challenging phases, especially when subsequent GMP compliance is required. To boost and support successful commercial introduction of biological…
Small Scale Escherichia Coli Cultivation Using BioBLU Single-Use and Reusable Vessels: A Comparative Study
In recent years, single-use bioreactors have gained more importance in animal and human cell culture. With its new line of BioBLU frigid-wall, stirred-tank, single-use vessels, Eppendorf offers premium solutions for microbial applications. In the following case study, reproducible process control was achieved with parallel-operated BioBLU 0.3f single-use and reusable glass vessels, both used in an Eppendorf DASbox® mini bioreactor system. Fermentation of Escherichia coli K12 led to very comparable results, thus proving tested single-use vessels to be an appropriate tool…
Continuous Processing and Integrated Facilities
Single-use technology has transformed the biomanufacturing industry, because single-use products provide significant savings over stainless steel infrastructure. Single-use technologies also create flexibility for biomanufacturers, enabling them to move from single-product processes to multiproduct facilities. As revolutionary and transformative as single-use technologies have been, the industry is on the cusp of another major shift, which comes with its own impressive benefits, but with some challenges as well. Continuous and Batch Processing Continuous processing has been used successfully for many years in…
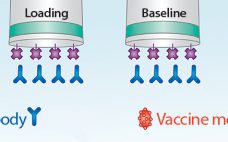
A Fast and High-Precision Influenza Vaccine Potency Assay
Fast and accurate determination of vaccine titer during manufacturing is important for understanding the performance of a development process and for scaling each process step. Although single radial immunodiffusion (SRID) has been the most commonly used technique for vaccine titer determination, it can be time-consuming and imprecise, requiring up to three days for results. The Pall ForteBio Octet® system offers a simple and direct method for measuring vaccine–antigen–antibody binding, capable of delivering high-precision analysis in as little as three hours.…